New Features: Event Queries, Targeted SDV, and more...
Release Date: October 20, 2017Release Date: October 20, 2017
We are pleased to bring you the following new features in this week’s release. See details about each feature’s enablement below.
EDC
timeDiff Function for Data Validation Rules
With this release, clinical programmers can build rules to return the difference between two times, using the function timeDiff(time1, time2). When clinical programmers build rules using this function, Vault EDC can validate that a given DateTime- or Time-_type _Item value falls within a given range, further automating the data validation process.
Definition Objects Read-Only when Published
This release extends the functionality of study publishing. After an Admin publishes the Study, all definition objects (except for Rules) become Read-Only. This feature provides a mechanism to control inadvertent changes to the study design outside of an expected study update.
Object Reuse in Studio
With object reuse, clinical programmers can manage and reuse standardized object definitions in a single Study and across multiple Studies in Studio. Clinical programmers can validate standards, make references to other objects, and override Labels to allow the use of objects as templates for other objects. With this feature, clinical programmers can develop a library-like collection of study design object definitions in Studio, saving time and increasing consistency.
System Rule Management in Studio
With this release, users can manage future date, mandatory (required), and range fields from an Item’s Properties panel, rather than through Admin > Business Admin. Users can also import and export system rules during casebook versioning. This provides the ability to maintain these data validation rules in Studio, simplifying the study development process and saving clinical programmers time.
Collapse & Expand Components in Design View
This feature allows users to collapse and expand all components in the Design view. Users can more easily navigate large visit schedules and form designs with less scrolling. Users are still able expand and collapse objects at an individual object level.
Updated Icons for Form Status
In this release, we updated the icons for Form status to be more visually consistent across different statuses. There is no change in behavior for which icons display on the casebook schedule. Each status still corresponds to an icon, and the icon only displays when a Form is in a given status.
Task Bar Performance Improvements
In this release, we updated the task bar’s count feature to improve performance. For users with access to multiple Studies, the task bar does not display any task counts until the user selects a single Study. After Study selection, the task bar counts display, and users can click into each type of tasks as they were able to previously.
Event Queries
Users with the ability to open manual queries can now create queries against an Event. Users can use Event queries to raise questions or get clarification about Event-level details, such as the Event date. To create an Event query, users with Create Query permission (CRAs and data managers) can select Open Query from the Event’s More Actions menu.
Users who answer queries (Investigators and CRCs) can do so from the casebook schedule, in the same way they would for an Item query.
Targeted SDV
With the introduction of Targeted SDV, a study sponsor can define SDV requirements for a subject in a Review Plan. Targeted SDV is extremely flexible and supports varying SDV requirements across all Items at the Event-_level. Sponsors can also configure Targeted SDV for some _Events to be very granular, drilling down to a specific Event, Form, Item Group, and Item combination. For each Item in the Targeted SDV Review Plan, there are three (3) modes available: Required, Optional, and Not Available. When an Item is Not Available, Vault EDC does not display an SDV checkbox for that Item in the UI. Sponsors can configure Targeted SDV plans at the Study- level for all Sites in that Study. Targeted SDV empowers sponsors to better define their review plans. Because clinical monitors now only need to SDV Items where it is required, those users can save time.
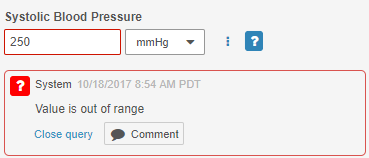
System-Generated Query UI Update
On system-generated Queries, the Reply button is now labeled Comment, and the color of the button is now gray. This label and styling change emphasizes that this button is for replying to a comment, rather than updating a queried Item value.
Sort the Casebook Schedule by Scheduled or Actual Date
On the casebook schedule, EDC users can choose to sort events in two different ways. Users can view the default casebook schedule with Events ordered by the Study schedule, and with unscheduled events at the bottom. Users can also toggle to sort Events sorted based on actual Event date recorded for the Events.
Reference Number for Repeating Forms
Repeating Forms are automatically assigned a reference number that displays in both form and table view, as well as in the form carousel. This number is now labeled as Reference #.
Vault Objects
Collapsible Page Sections
This feature introduces several changes that significantly improve Vault’s performance when loading an object record detail page:
- All sections except the first details section appear in a “collapsed” state by default.
- Users can expand/collapse individual sections by clicking on the section heading.
- Jumping to a section using the Navigation Panel on the right of the page automatically expands that section.
With this feature, we’ve also changed how related records and documents display within a section. In past releases, we showed 25 items and allowed users to expand to show all items within the section. In this release, we show 20 items at a time and allow users to page through the items.
Page Links
Admins can create page link URLs that navigate users to custom page destinations, such as a custom user interface for simplifying data entry into an object for tracking events. At runtime, page links direct users to the URL defined by the link as opposed to the standard object record detail page. Admins can create Page Links with custom URL destinations for the following object record actions:
- Create
- View
- Edit
- Copy
Page link URLs can be internal or external. Vault loads external pages in an iFrame.
UI Update for Object Record Permission Details
In this release, we’ve modified the UI to allow Admins to edit object permissions at the profile and field level from the Users & Groups > Permission Sets > Objects > [Object] details page. This update uses the same design as Vault’s object page layout providing consistency and improved usability for users.
The object record permission details page has an updated layout. You can now edit object level permissions (Read, Create, Edit, Delete) from the object record permission details page without the enablement of object types.
System-Managed Name Enhancements
Today, Admins can configure Vault to automatically generate object record names by enabling the System manages field value for the Name field. This feature enhances the Value Format to support field tokens, in addition to the literal strings and auto-numbering tokens supported today.
Field tokens can come from the same object or from objects related through outbound relationships. Vault supports tokens for the following field types:
- Text
- Number
- Picklist (single-value only)
With this release, Admins can also change the Value Format and Starting Number for standard objects with System manages field value enabled.
Object Reference Constraints Enhancement
With this enhancement, Admins can apply object reference constraints based on lifecycle state. This allows users to view and select from a list of different reference records in different lifecycle states.
Lifecycle & Workflow
Workflow Task Due Dates Based on Object Fields
When configuring object workflows, Admins can now set task due dates based on date field values from the object record, with or without an offset value. This enhancement simplifies data entry and reduces the chance of users entering dates incorrectly by leveraging values that already exist on the object record. For example, an organization that needs to review Product records every six (6) months could set due dates based on Last Product Review Date plus 180 days.
Reporting
Reorder Report Objects
When using report types that include multiple up objects, users can now control the order of objects. Users can access this option through the existing Edit Fields dialog. Prior to this feature, Vault always displayed the primary object in the leftmost columns and sorted other objects alphabetically.
This feature also allows users to sort report results on any field from an up object that the primary object references. For example, in a report showing marketing campaigns with documents, users could sort by the Product Approval Date, a field that belongs to the Product object referenced by the Marketing Campaign object.
Vault Loader & Configuration Migration
Vault Configuration Report: Suppress Empty Results
The new Suppress Empty Results option allows users to hide report tabs with no results when generating the Vault Configuration Report.
Vault Compare Permission
Admins can now grant or limit access to the Vault Compare feature using custom security profiles and the new Environment: Vault Comparison permission.
New Supported Component Types
The following component types are now supported for Configuration Migration Packages, Vault Compare, and Vault Configuration Report:
- Formatted Output Template
- Document Matching Sharing Rule
Action Level Security (ALS)
Vault Actions are configured custom actions in Vault that can be executed on an object record. Action Level Security (ALS) provides a unified way to secure those actions. With ALS, you can configure permissions at the Security Profile level to apply to actions across all states, and at the Atomic Security level to provide more granular control by state and role.
ALS: Atomic Security for Actions
With Atomic Security for Actions, you can secure object actions and lifecycle user actions at the role level. This allows you to control which object actions or lifecycle user actions are hidden, viewable (visible but grayed out), or executable (visible and clickable) for a specific role.
Security settings for a given action can vary by lifecycle state, and similar to Atomic Security for object fields, you can override the defaults for certain application roles.
ALS: Profile Security for Actions
When you configure object actions, you can secure those actions on the User Profile level through permissions sets.
For any object with one or more object actions enabled, Vault displays the Object Action Permission section on an object record’s Permissions page. This section allows you to assign View and Execute permissions on the action.
Vault hides any actions without any set permissions at the User Profile level from users assigned to the profile. Actions with the View permission appear in the Action menu of an object record, but users cannot execute them. The Execute permission allows users to view and execute an action.
ALS: Object Actions
With this release, your organization can work with Veeva Services to define actions for one or more objects allowing users to perform actions on those object records. Objects now have a new Actions tab that displays a list of securable custom object actions. Custom actions are configurable instances of a Vault Action. Additionally, an Admin can secure the action using ALS to determine if the action is hidden, view-only, or executable at runtime.
Note that Admins must contact Veeva Services to configure Vault Actions at this time. Additionally, Standard Actions will not appear in the Actions tab at this time.
Feature Enablement Details
| Feature | Enablement | Application |
|---|---|---|
| EDC | ||
| timeDiff Function for Data Validation Rules | Auto-On | EDC |
| Definition Objects Read-Only when Published | Auto-On | EDC |
| Object Reuse in Studio | Configuration | EDC |
| System Rule Management in Studio | Auto-On | EDC |
| Collapse & Expand Components in Design View | Auto-On | EDC |
| Updated Icons for Form Status | Auto-On | EDC |
| Task Bar Performance Improvements | Auto-On | EDC |
| Event Queries | Auto-On | EDC |
| Targeted SDV | Configuration | EDC |
| System-Generated Query UI Update | Auto-On | EDC |
| Sort the Casebook Schedule by Scheduled or Actual Date | Auto-On | EDC |
| Reference Number for Repeating Forms | Auto-On | EDC |
| Vault Objects | ||
| Collapsible Page Sections | Auto-On | Platform |
| Page Links | Configuration | Platform |
| UI Update for Object Record Permission Details | Auto-On | Platform |
| System Managed Name Enhancements | Configuration | Platform |
| Object Reference Constraints Enhancements | Configuration | Platform |
| Lifecycle & Workflow | ||
| Workflow Task Due Dates Based on Object Fields | Configuration | Platform |
| Reporting | ||
| Reorder Report Objects | Auto-On | Platform |
| Vault Loader & Configuration Migration | ||
| Vault Configuration Report: Suppress Empty Results | Auto-On | Platform |
| Vault Compare Permission | Configuration | Platform |
| Bulk Document Relationship Creation using External ID | Auto-On | Platform |
| New Supported Component Types | Auto-On | Platform |
| Action Level Security (ALS) | ||
| ALS: Atomic Security for Actions | Auto-On | Platform |
| ALS: Profile Security for Actions | Configuration | Platform |
| ALS: Object Actions | Support | Platform |
Enablement Legend
See the following explanations for feature enablement options:
| Enablement | Description |
|---|---|
| Auto-on | Automatically activated and no configuration is required before using the feature; note that in some cases, a new feature is dependent on another feature that must be enabled or configured. |
| Admin Checkbox | Admins must turn on the feature with an Admin checkbox. Note that some “Auto-On” features have a checkbox setting that hides the feature; these will show “Auto-On.” |
| Configuration | Admins must configure the feature (separately from an Admin checkbox) before it is available to use or is active; for example, a Clinical Programmer must create an Item Definition of a certain new Item Type. |
| Support | On/off option controlled by Support. |