Exporting PDFs
In Veeva EDC, you can export your subject’s Casebook to a PDF to print or to use as a reference.
Prerequisites
Users with the CDMS Clinical Research Coordinator, CDMS Data Manager, and CDMS Lead Data Manager can perform the actions described below by default. If your organization uses user-defined Study Roles, your role must grant the following permissions:
| Type | Permission Label | Controls |
|---|---|---|
| Functional Permission | Generate Blank PDF | Ability to export blank PDFs |
| Functional Permission | Generate Detail PDF | Ability to export detail PDFs |
If your Study contains restricted data, you must have the Restricted Data Access permission to view it.
Learn more about Study Roles.
Available PDFs
You can export two (2) different types of PDFs for Forms and Casebooks in your Study.
- Blank PDFs that have no data.
- Detail PDFs that have all entered study data.
For Detail PDFs, you can choose whether to include queries, audit trail information, or both.
At the Form-level, the PDF includes the Form, as well as the name of the Study, Event, and Form in a header. If you exported a Detail PDF with queries, the PDF also includes sections for audit information and queries.
At the Casebook-level, the PDF includes all Forms in the Casebook, ordered by Event, as well as Study and Subject information in the header. The Event and Form records inside the casebook are bookmarked in the PDF with an option to view By Visit or By Form through the enhanced Detail PDF feature.
PDF Language: Detail and Blank PDFs are available in the language that is set on your user profile. PDFs are translatable, however, only standard text will be translated. Configured labels or entered text will not be translated.
Detail PDF
Detail PDFs are available for both forms and entire casebooks. Note that blank spaces or special characters that are included in site names or numbers will be replaced with underscores (_) for file and folder names.
If your Study was created before the 19R3 release, you may need to enable this feature in order to use Detail PDFs.
File Size Limit: Detail PDFs are limited to 4GB in size.
Exporting PDFs
Exporting PDFs from the Review Tab
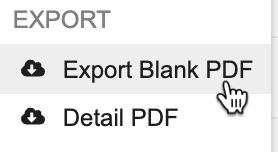
Exporting Blank PDFs
To export a Blank PDF from the Review tab:
- Navigate to the Study and Site containing the Subject you want to generate a PDF from.
-
Optional: To export a Subject-level PDF including all Forms associated with the Subject’s__ Casebook, on the Subject’s page, click the More Actions menu.
-
Optional: To export a PDF of a single Form, navigate to the Form and click the More Actions menu next to the Form name.

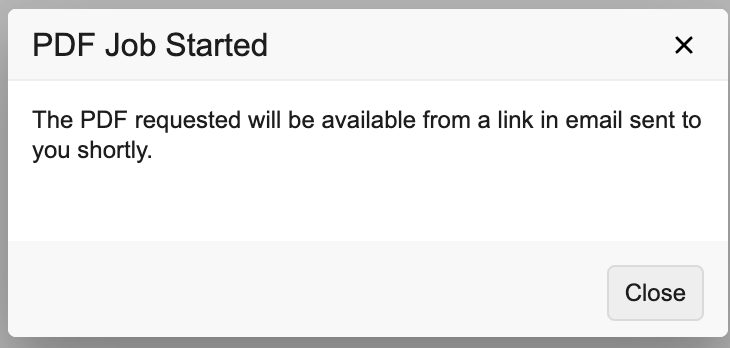
- Vault begins generating your PDF. The system automatically downloads Form-level PDFs. When generation of Subject-level PDFs is finished, you will receive an email with a link to download the PDF.
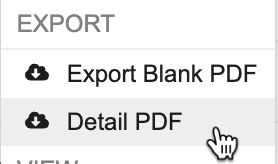
Exporting Detail PDFs
To export a Detail PDF from the Review tab:
- Navigate to the Study and Site containing the Subject you want to generate a PDF from.
-
Optional: To export a Subject-level PDF including all Forms associated with the Subject’s Casebook, on the Subject’s page, click the More Actions menu.

-
Optional: To export a PDF of a single Form, navigate to the Form and click the More Actions menu next to the Form name.

- Optional: Select Include Queries to include all the queries associated with the Casebook.
-
Optional: Select Include Audit Trails to include audit trail descriptions associated with data changes in the Casebook.
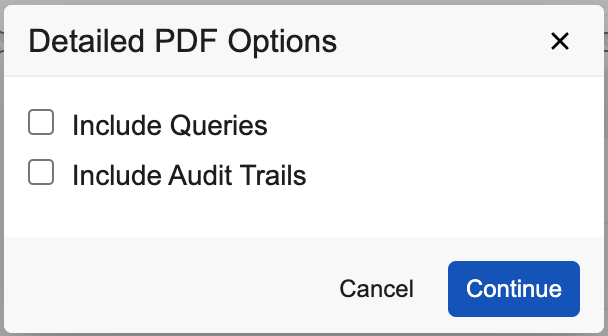
- Click Continue.
- Vault begins generating your PDF. The system automatically downloads Form-level PDFs. When generation of Subject-level PDFs is finished, you will receive an email with a link to download the PDF.

Exporting PDFs for All Subjects in a Site
You can export a Detail PDF or a Detail PDF including queries and audit trail from the EDC Tools > Jobs tab. You can choose to export PDFs for a single Site, a selection of Sites, or all Sites in your Study (Closeout PDF). Note that this job is only available to users with the EDC Tools Tab permission. See Job Management for details.
Sort By Visit & By Form
You can view Events by visit by selecting By Visit in the PDF bookmark section. In this view, you will see all subject events ordered by study design and containing all forms corresponding to that event. Use the By Form bookmark to organize forms alphabetically.
Deleted Data in PDFs
If you select Include Audit Trail when generating your PDF, your PDF files include deleted data in the Deleted Data Audit Trail section of the file’s appendix. This includes any data that was deleted as part of the following processes:
- Retrospective amendment with destructive design changes
- Site reset an event or form which was marked for removal
- Site reset a repeating event
- Site removed row in a repeating item group
- Site removed the form linking by deleting a linked form
- Query deletions, undo action in Quick Queries*
- Unscheduled Event is deleted
* The Undo action in Quick Queries is captured as a delete action. It’s included in the audit trail for the queried Item or Event. It’s only included in the Deleted Data Audit Trail section if the queried Item or Event is also deleted.
Only users with access to restricted forms (users whose study role grants the Restricted Data Access permission) will see the Deleted Data Audit Trail. If the user who generated the file doesn’t have restricted data access, and there are deletions of restricted data, then Vault includes a note at the end of the PDF:
Note: The Deleted Data Audit Trail was not included in this file because the user who generated it did not have restricted data access. To include this data, please generate the file again as a user with restricted data access.
Replace Non-Permitted Characters from PDF File & Folder Names
Vault removes non-permitted characters from file and folder names in your PDF exports and replaces them with an underscore (_).
Your organization can choose to instead replace these characters with a hyphen (-).
Enablement: If your vault was created prior to the 25R2 release (August 2025), you must contact Veeva Support to use hyphens instead of underscores. If your vault was created after the 25R2 release, your vault will use hyphens to replace non-permitted characters by default.
When generating the PDFs, any of the following non-permitted characters will be replaced with a hyphen or underscore, depending on configuration:
- Periods (.)
- Whitespaces
- Exclamation points (!)
- At signs (@)
- Pound signs (#)
- Dollar signs ($)
- Carets (^)
- Percent signs (%)
- Asterisks (*)
- Commas (,)
-
Pipes ( ) - Plus signs (+)
- Quotation marks (“)
- Tildes (~)
- Less than or greater than, angle brackets (< or >)
- Curly brackets ({ or })
- Forward or back slashes (/ or )
- Equals sign (=)
- Brackets ([ or ])
- Underscores (_), if configured to replace with a hyphen
When deploying a study, if the user chooses to include Detail PDFs in the deployment file, the file names will not include the replacement hyphens. These files are for internal record keeping and not for regulatory review.